E-Methanol – A Renewable Fuel for Hard-to-Decarbonise Sectors
What is E-Methanol?
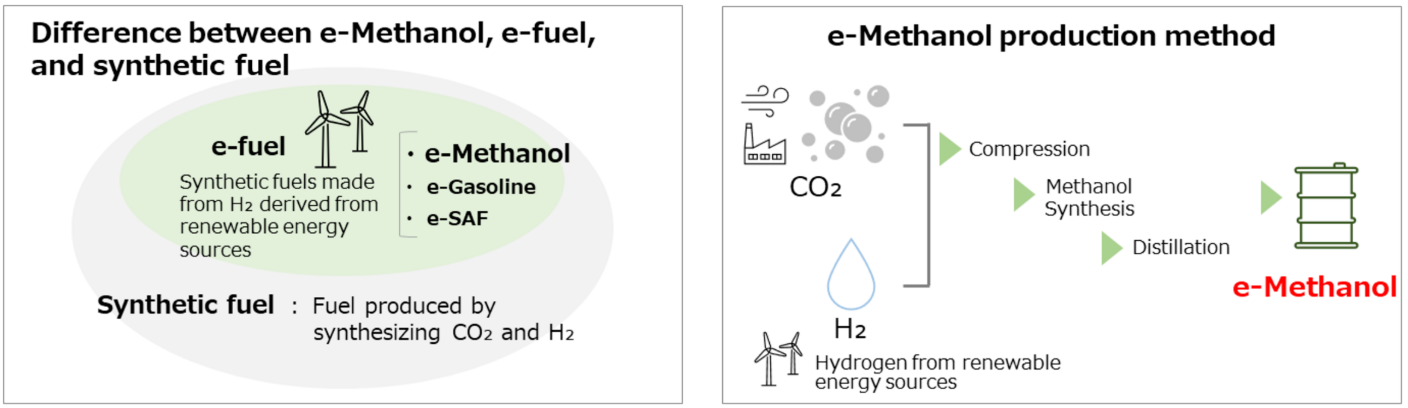
E-methanol (electro-methanol) is a synthetic, renewable liquid fuel made by combining green hydrogen (from water electrolysis powered by renewable energy) and captured carbon dioxide (CO₂).
How It’s Made:
-
Green Hydrogen Production – Renewable energy (solar/wind) splits water into hydrogen and oxygen.
-
CO₂ Capture – CO₂ is sourced from industrial emissions or direct air capture.
-
Methanol Synthesis – Hydrogen and CO₂ are combined in a catalytic reactor to form methanol.
Why It’s Important:
-
Decarbonisation – Offers low/zero-emission fuel for sectors like shipping and aviation.
-
Infrastructure Compatibility – Can use existing storage and fuel systems.
-
Versatile Applications – Usable in transport, aviation, and chemical industries.
-
Carbon Neutral Potential – If produced with renewable energy and captured CO₂, it can be nearly emission-free.
Challenges:
-
High Cost – Expensive compared to conventional methanol due to costly technology and renewable power.
-
Scalability – Needs major infrastructure and investment to meet global demand.
-
Public Awareness – Understanding and acceptance of synthetic fuels are still limited.
Example:
-
Kassø Facility (Denmark) – One of the world’s first and largest commercial e-methanol plants, supplying fuel for the shipping sector.
E-methanol is a promising clean fuel alternative for hard-to-electrify sectors, with potential to drive global decarbonisation, though challenges in cost and scale remain.
